N-Methylation of Peptides: A New Perspective in Medicinal Chemistry
18-Jul-2008
Accounts of Chemical Research, 2008, DOI: 10.1021/ar8000603, published on 18.07.2008
http://pubs.acs.org , online article
http://pubs.acs.org , online article
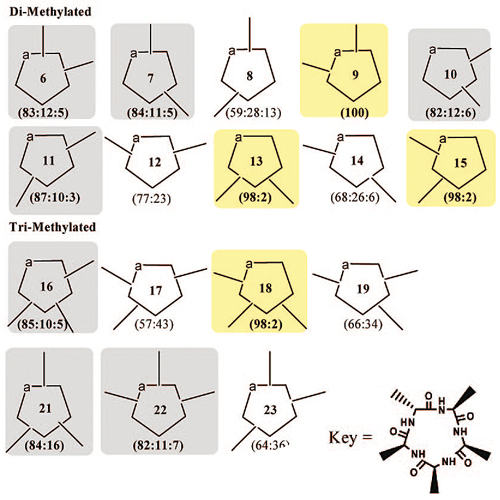
The potential of peptides as drug candidates is limited by their poor pharmacokinetic properties. Many peptides have a short half-life in vivo and a lack of oral availability. Inspired by the excellent pharmacokinetic profile of cyclosporine, a natural, multiply N-methylated cyclic peptide, we envisioned multiple N-methylation as a promising way to rationally improve key pharmacokinetic characteristics. In this Account, we summarize our efforts toward modulating the properties of peptides by multiple N-methylation. As a first step, we simplified the synthesis of N-methylated amino acids in solution, by employing very mild conditions that could be tolerated by the diverse protecting groups required when working with naturally occurring amino acids. We also report the rapid and inexpensive syntheses of N-methylated peptides on a solid support; this facilitated the N-methyl scanning of bioactive peptides. Because of a lack of information regarding the conformational behavior of multiply N-methylated peptides, a complete library of N-methylated cyclic alanine pentapeptides was synthesized. The library provided valuable insight into the conformational modulation of cyclic peptides by N-methylation. This information is extremely valuable for the design of bioactive peptides and spatial screening of cyclic N-methylated peptides. To demonstrate the applicability of N-methylation to highly active but poorly bioavailable peptides, we performed a full N-methyl scan of the cyclopeptidic somatostatin analog cyclo(-PFwKTF-), known as the Veber-Hirschmann peptide. We show here for the first time that the simple approach of multiple N-methylation can drastically improve the metabolic stability and intestinal permeability of peptides, for example, resulting in 10% oral bioavailability for a tri-N-methylated Veber-Hirschmann peptide analog. In addition, we also describe a designed approach to N-methylated peptide library synthesis, which can accelerate the screening of N-methylated bioactive peptides. Finally, we find that multiple N-methylation of a cyclic hexapeptide integrin antagonist of GPIIb-IIIa (RIIb_3 integrin), cyclo(-GRGDfL-), increases the selectivity of this peptide toward different integrin subtypes. This result demonstrates the utility of multiple N-methylation in elucidating the bioactive conformation of peptides.