sNASP and ASF1A function through both competitive and compatible modes of histone binding
05-Oct-2016
Nucl. Acids Res., doi: 10.1093/nar/gkw892
Nucl. Acids Res., online article
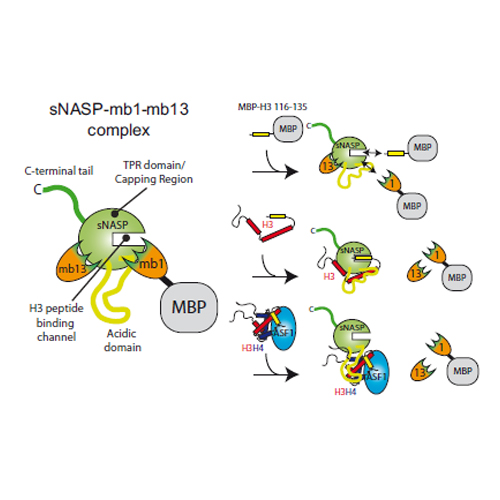
Histone chaperones are proteins that interact with histones to regulate the thermodynamic process of nucleosome assembly. sNASP and ASF1 are conserved histone chaperones that interact with histones H3 and H4 and are found in a multi-chaperoning complex in vivo. Previously we identified a short peptide motif within H3 that binds to the TPR domain of sNASP with nanomolar affinity. Interestingly, this peptide motif is sequestered within the known ASF1–H3–H4 interface, raising the question of how these two proteins are found in complex together with histones when they share the same binding site. Here, we show that sNASP contains at least two additional histone interaction sites that, unlike the TPR–H3 peptide interaction, are compatible with ASF1A binding. These surfaces allow ASF1A to form a quaternary complex with both sNASP and H3–H4. Furthermore, we demonstrate that sNASP makes a specific complex with H3 on its own in vitro, but not with H4, suggesting that it could work upstream of ASF1A. Further, we show that sNASP and ASF1A are capable of folding an H3–H4 dimer in vitro under native conditions. These findings reveal a network of binding events that may promote the entry of histones H3 and H4 into the nucleosome assembly pathway.