Sortase-catalyzed in vitro functionalization of a HER2-specific recombinant Fab for tumor targeting of the plant cytotoxin gelonin
09-Dec-2013
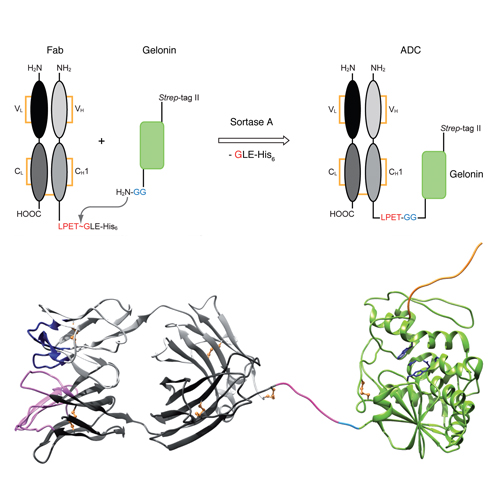
We report on the preparation of a new type of immunotoxin via in vitro ligation of the αHer2 antigen binding fragment (Fab) of the clinically-validated antibody trastuzumab to the plant toxin gelonin, employing catalysis by the bacterial enzyme sortase A (SrtA). The αHer2 Fab was fused with the extended SrtA recognition motif LPET↓GLEH6 at the C-terminus of its heavy chain, thereby preventing interference with antigen binding, while the toxin was equipped with a Gly2 sequence at its N-terminus, distant to the catalytically active site in the C-terminal region. Site-specific in vitro transpeptidation led to a novel antibody-toxin conjugate wherein gelonin had effectively replaced the Fc region of a conventional (monomerized) immunoglobulin. After optimization of reaction conditions and incubation time, the resulting Fab-Gelonin ligation product was purified to homogeneity in a two-step procedure by means of Strep-Tactin affinity chromatography—utilizing the Strep-tag II appended to gelonin—and size exclusion chromatography. Binding activity of the immunotoxin for the Her2 ectodomain was indistinguishable from the unligated Fab as measured by real-time surface plasmon resonance spectroscopy. Specific cytotoxic potency of Fab-Gelonin was demonstrated against two Her2-positive cell lines, resulting in EC50 values of ~1 nM or lower, indicating a 1000-fold enhanced cell-killing activity compared with gelonin itself. Thus, our strategy provides a convenient route to the modular construction of functional immunotoxins from Fabs of established tumor-specific antibodies with gelonin or related proteotoxins, also avoiding the elevated biosafety levels that would be mandatory for the direct biotechnological preparation of corresponding fusion proteins.